*See a properly formatted version here.
INTRODUCTION
There are three buttons in front of you, labeled “butter,” “brown sugar,” and “chocolate.” With one press of them all, the air is infused with the rich, creamy foundation of butter, the caramel-like sweetness of brown sugar, and the slightly bitter aroma of chocolate.
This may sound familiar to those who have visited the Museum of Food and Drink in New York City, where an elephant-trunk-shaped machine sits and produces those smells. The truth is, you are smelling chemicals, such as methyl cyclopentenone (found commonly in food containing cooked sugar and maple syrup),1 and diacetyl, which has a strong buttery vanilla cream smell.2 The machine emulates those used by international flavor houses responsible for blending flavor chemicals into processed foods, such as International Flavors & Fragrances (IFF), Firmenich, and Givaudan.3 These companies carefully blend artistry and precision to recreate all the flavors we love.
Behind every flavor is a world few of us ever think about. The concept of “natural and artificial flavors” is ubiquitous, yet very few people consider what these words mean. Regulations for these flavors also often blur the lines between “natural” and “artificial” flavors. As global demand for more sustainable flavorings grows, a deeper understanding of the regulatory framework becomes critical for both consumers and producers.
This paper first delves into the scientific understanding of food flavorings, followed by an examination of the current regulatory framework under the U.S. Federal Food, Drug, and Cosmetic Act (FDCA). It also explores the issues and gaps in regulating flavors, particularly in FDA enforcement mechanisms, challenges in compliance and labeling, and emerging concerns in the industry (especially those related to safety and allergens). This paper ultimately makes recommendations for closing current loopholes in the GRAS system, controlling quantities of toxic flavors, addressing vague labeling concerns, and enhancing future FDA enforcement actions. Examining the regulatory framework for flavors is a matter of maintaining compliance, preserving public trust, and protecting consumers in an increasingly complex food landscape.
I. SCIENTIFIC UNDERSTANDING OF FOOD FLAVORS
A. THE CHEMISTRY OF FLAVORS AND EXTRACTS
Flavors are used extensively in food production.4 While flavors like “chocolate” seem straightforward, they are chemically complex as they interact with our receptors to produce taste and aroma.5 A pineapple tastes the way it does because it consists of sweet and sour flavors existing at different levels of potency—one of its components is so potent that people can detect it even in tiny amounts, like a few grains of sugar in an Olympic-size pool.6
These chemicals are difficult to generalize using a single term since even a simple flavor can contain thousands of components.7 For example, when making apple pies, bakers often add cinnamon and nutmeg to enhance the taste of cooked apples.8 A product like Tastykake glazed apple pie lists these spices on its label along with “natural and artificial flavors.”9 These flavorings could also include natural vanilla extracts and artificial butter flavor, but since the label only lists “natural and artificial flavors,” consumers are not informed about the exact ingredients.10
B. NATURAL V. SYNTHETIC EXTRACTS
Substances used in creating flavoring can either be naturally derived or artificially produced and can exist as either a single chemical like vanillin or complex mixtures created by food chemists.11 Generally, natural flavors are derived from nature and function predominantly for flavor instead of nutrition.12 Among those, flavors can be “nature-derived” from natural fruits, herbs, and spices, and may or may not include chemically processed compounds classified as natural under regulatory definitions.13 For example, a lemon flavor can be named either “Natural Lemon Flavor FTNF,” meaning all of its flavor components must be sourced “from the named food” (lemon), or “Natural Lemon Flavor WONF,” which means the flavor must come “with only natural flavors” or from any amount of a natural derivative from lemon.14 These flavors can also be industrial recreations of natural molecules known as “nature-identical” compounds, or “processing aids” such as solvents or emulsifiers to generate taste consistency.15
Consumers often prefer natural flavors and assume negatively about “artificial” ones. Unlike natural flavors, artificial flavors are not derived from nature.16 Artificial flavors are often single compounds synthesized to mimic the taste of natural ingredients, offering a cost-effective solution for manufacturers who need not hunt down natural sources to recreate a taste.17 For example, the same chemical compound of vanillin can be produced from paper pulp instead of vanilla beans.18 Oftentimes, hybrid flavors combine natural and artificial elements to achieve a balanced taste profile. A product may be labeled “Natural & Artificial Lemon Flavor” or “Natural & Artificial Lemon-Type Flavor”, which can be a combination of flavors from lemon only, lemon- derivative ingredients, and synthetic compounds.19 These hybrids allow manufacturers to achieve flavor stability for products on store shelves.20
C. USAGE IN FOOD PRODUCTS: BALANCING SAFETY AND INNOVATION
Through improvements in chemical extractions21 and focuses on carbon footprint minimization,22 flavor companies can create trade-secret-protected blends of natural and synthetic substances, which often consist of over 100 ingredients, tailored to manufacturers’ specific needs.23 While using these blends simplifies the manufacturing process, balancing such innovation with safety regulations became more complicated.
II. FDA’S REGULATORY FRAMEWORK AND ENFORCEMENT
A. FOOD FLAVOR REGULATION (FDCA, FOOD ADDITIVE AMENDMENTS, GRAS SYSTEM)
In the U.S., the Food and Drug Administration (FDA) and the U.S. Department of Agriculture (USDA) regulate food flavors.24 While the USDA oversees flavor safety and labeling in meat, egg, poultry, and fish products, the FDA does so for packaged food and beverages before food companies begin utilizing them.25 The FDCA defines “food” to include raw ingredients and empowers the FDA to oversee food safety.26 Part of such authority includes ensuring that food and its additives are not adulterated with harmful substances.27 This means that there must be a “reasonable certainty” that the food and added substances will not cause cancer development through its intended use.28
To regulate this, the FDA employs guidance documents and petition processes. For instance, in response to IFF’s failure to warn about damages to respiratory functions from diacetyl exposure, the FDA reassessed the safety status of diacetyl and published recommendations for recategorizing unsafe flavors.29 The FDA has also implemented regulations established by broader legislation passed by Congress. The NLEA (Nutrition Labeling and Education Act) of 1990 gave the FDA authority to mandate disclosure of particular nutrition information.30 The specific requirement that food labels list any “artificial flavor” or “natural flavor” used in the product is codified in 21 C.F.R. § 101.22.31 The FALCPA (Food Allergen Labeling and Consumer Protection Act) of 2004 also tasks the FDA with mandating labeling of major allergens, including those in flavors.32
Flavors are classified as food additives,33 and are subject to premarket approval unless deemed GRAS.34 In other words, there are two pathways through which a new flavor can enter the food supply, the default being the FDA’s premarket approval. If a company wants to introduce a new flavor, it submits a petition demonstrating that it is safe under proposed uses.35 Subsequently, the FDA assesses the petition through scientific review, evaluating the identity, probable exposure, and limitations of conditions of use.36 As part of this review, the public can also give their input.37 If the flavor is approved, companies are allowed to use the new flavor while it is continuously regulated under the Food Additives Amendment of 1958; in contrast, if the petition is rejected, companies may not utilize the new flavor.38
However, since this premarket approval process is time-consuming, stringent, and costly, many companies turn to the alternative GRAS system. Since 1997, the GRAS (Generally Recognized as Safe) system has allowed substances to bypass the FDA’s premarket approval process through a voluntary notification system.39 Congress “exempted” food substances with established safety, which is proven by long history of uses or scientific confirmation.40 This criteria has expanded to food additives as well.41 When a company submits a GRAS notice, the FDA reviews the documentation provided but does not independently evaluate the safety of the substance.42 Alternatively, through “Secret GRAS,” companies can even self-certify an ingredient as “generally safe” and use it in food without notifying the FDA.43
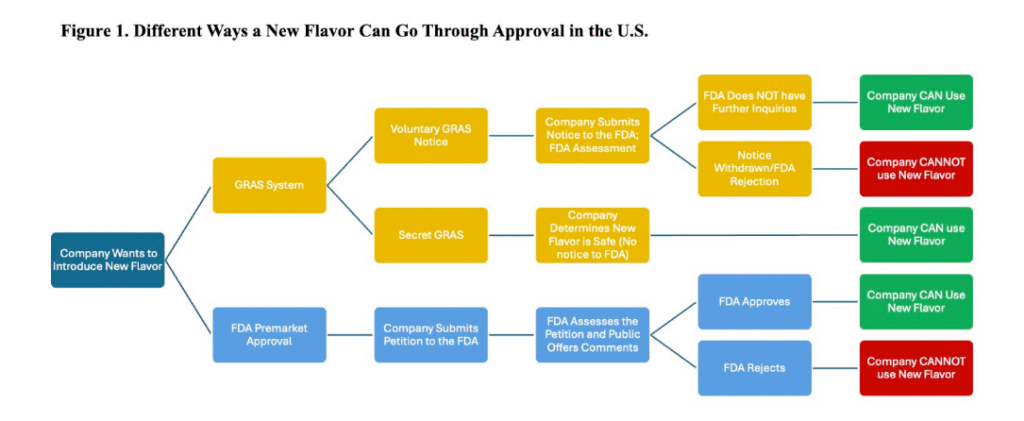
Currently, out of the 10,000 chemicals in the U.S. food supply, over 3,000 go through the GRAS system and skip the FDA approval process.44 This shortcut through GRAS has expanded beyond household ingredients such as vinegar and baking powder.45 The GRAS system is much less rigorous compared to a formal food additive petition, and companies are also given more discretion to determine what ingredients can be “GRAS” without being on the FDA’s radar at all.46
B. BANNED AND UNSAFE FLAVORS
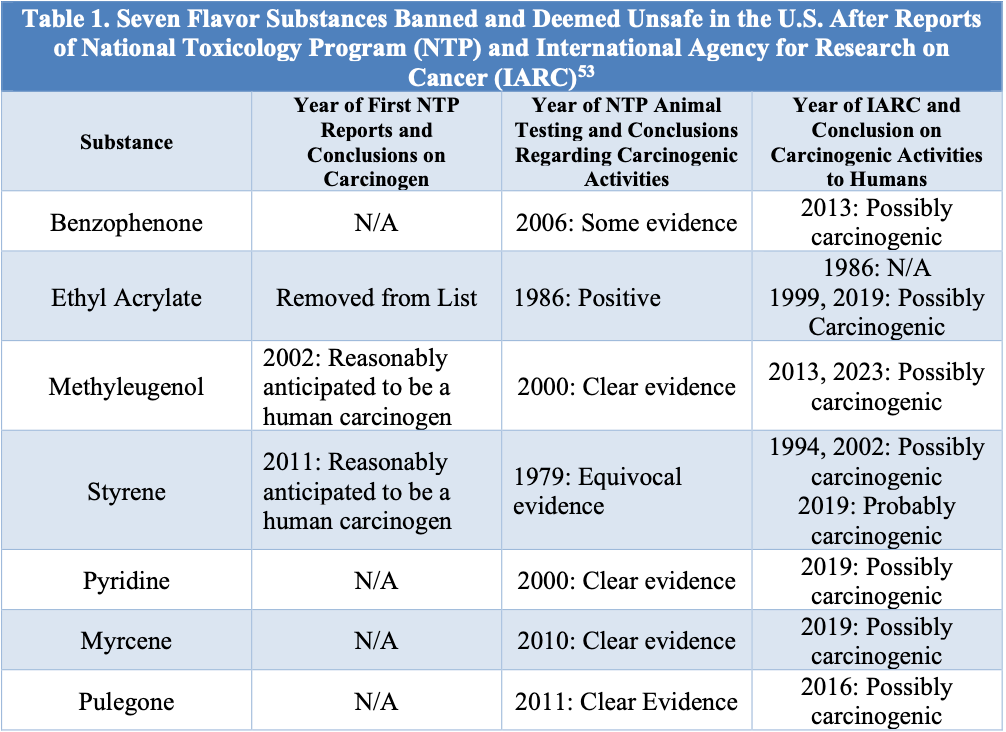
The FDA bans harmful substances, which include both FDA-approved additives and those introduced through the GRAS system.47 In 2018, after evidence of carcinogenic potential arose in animal studies, the FDA banned seven synthetic flavors: benzophenone, ethyl acrylate, methyleugenol, myrcene, pulegone, pyridine, and styrene (see Table 1).48 Through the Delaney Clause of the FDCA, the FDA is required to find these substances as unsafe and not approve the use of any food additives with known quality to induce cancer at any dosage level.49 Recently, the FDA banned the use of BVO (brominated vegetable oil) in sports drinks and sodas after finding BVO, which contains bromine (used in fire retardants), in beverages to maintain flavor consistency.50 These synthetic flavors have not been used in large doses or caused high risks of cancer, but the FDA still revoked them to protect public health.51 However, even though many of these substances had decade-long linkages to cancer, their actual revocations often came years after the initial petitions to ban.52
See footnote53
C. LABELING AND CONSUMER PROTECTION
1. LABELING OF FLAVORINGS
Natural flavors, as defined in 21 C.F.R. § 101.22, refer to substances like essential oils or extracts derived from natural sources, such as fruits, herbs, or meat, used primarily for flavoring rather than nutritional purposes.54 To break down this verbose labeling rule, consider a blueberry muffin snack. When the characterizing ingredient fully flavors the snack, the snack can simply be labeled as “blueberry muffin.”55 On the contrary, if an ingredient only functions to supplement a food’s natural taste without being the primary flavor, it is simply listed as “natural flavors.”56 If the snack does not include the characterizing ingredient (i.e., blueberries) but contains a flavor ingredient derived naturally from the simulated food (i.e., blueberry flavors derived from blueberries), the snack must be labeled as “natural blueberry flavor.”57 Additionally, if the snack contains a flavor ingredient sourced from the simulated food (i.e., blueberry flavor derived from blueberries) and other flavors that supplement the characterizing flavor, the label must reflect such combination as “natural blueberry flavor, with other natural flavors.”58
Meanwhile, “artificial flavors” labels can consist of a mixture of any of the above disclosures—the blueberry muffins would typically be labeled as “Naturally and Artificially Flavored Blueberry Muffin.”59 When the natural flavor is derived from a different source than the expected characterizing flavor, such as using grape-derived flavors in a blueberry muffin, the flavor must be labeled either as “blueberry muffin with natural grape flavor” or as “artificial flavor.”60 Furthermore, if the product’s packaging shows images or names of a specific food item representing the main flavor, yet the flavor is derived from natural flavors, the product must follow the above labeling requirements.61 However, if the flavor comes mainly from the actual food ingredient, the presence of natural flavors only needs to be indicated in the ingredients list without supplemental labeling details on the main display panel.62
2. MISBRANDING
The FDA defines “misbranding” as false or misleading labeling, which includes omissions or incorrect statements about the product’s ingredients, origins, or intended use.63 Many disputes surrounding flavor misbranding have been surfacing. In Lima v. Post Consumer Brands, the plaintiffs claimed the packaging of Honey Bunches of Oats, which featured honey imagery when the cereal was mostly sweetened with sugar and syrup, was misleading.64 In Viggiano v. Hansen Nat. Corp, the plaintiff claimed that Hansen’s diet sodas were misleadingly labeled as containing “all natural flavors,” despite the inclusion of synthetic sucralose.65 Target’s Market Pantry fruit punch-flavored water enhancer and MiO water enhancers were also under scrutiny for labeling its product with “natural flavor with other natural flavors” despite containing synthetic malic acid.66
Additionally, companies are not required to label what their “natural flavors” consist of, unless the ingredients include common allergens such as shellfish and tree nuts.67 There are also other synthetic flavor substances known as “adjuvants” not traditionally included in “artificial” or “natural” flavors.68 These adjuvants are usually only used in minimum quantity needed to generate the intended taste effect compliant with good manufacturing practices.69 Some have use limitations, such as Angola weed and artichoke leaves, which can only be used in alcoholic beverages.70 These additional classification nuances further compound the issue of flavor misbranding and the lack of transparency.71
D. FDA ENFORCEMENT ACTIONS
As the first line of action to ensure safety, the FDA issues warning letters to companies violating regulations, such as improper labeling or unauthorized use of food flavors, and demands corrective actions. For Kramer’s Jalapeno Pepper Jam, the FDA raised concerns about the declared amount of sugar on the label, which stated a mere “1g” despite sugar being the most predominant ingredient.72 Similarly, the FDA warned Lipari Foods Operating Company LLC that its labels failed to declare major food allergens like cashew, and that it implemented no preventive controls.73 To follow up, the FDA can seize unapproved, adulterated, misbranded, or unsafe flavors, such as tartrazine.74 The FDA can collaborate with companies to initiate voluntary or mandatory recalls of these flavors based on the level of risks to consumers, as seen with the reformulation and recalls of BVO.75 Import alerts or refuse entry have also been issued for products containing unapproved flavors.76
For repeated non-compliance, the FDA sometimes collaborates with the Department of Justice (DOJ) to pursue legal actions, resulting in civil penalties, criminal prosecutions, and injunctions. Many of these enforcement actions target “food frauds,” such as when companies dilute extra-virgin olive oil with cheaper vegetable oil, or brand their food as “made of purely honey and maple syrup” while mixing in cheaper cane sugar to lower production costs.77 For example, Valley Process has previously been barred by a court-ordered consent degree from operating due to its adulterated grape juice containing arsenic and patulin; a seafood company and cheese company have also been fined, put on probation, had their owners sent to prison for misbranding their products.78 The overall goal of these actions are to protect consumers from being cheated as well as the general health of the public.
III. EMERGING ISSUES AND GAPS IN REGULATING FLAVORS
A. GRAS LOOPHOLES
The GRAS system allows industry self-regulation to override public health, and even when the FDA is notified of a GRAS determination, it cannot conduct its own independent review.79 Initially, the GRAS exemption was designed for household ingredients, but it has evolved into a loophole, allowing companies to introduce unknown chemicals without FDA approval or public scrutiny.80
The Food Additives Amendment of 1958 defines GRAS substances as those “generally recognized as safe” among experts based on published scientific data, but there are ambiguities regarding which scientific procedures and experts are qualified to make determinations. Consumers assume the FDA rigorously reviews the safety of flavors through scientific reviews. However, in reality, many food companies rely on trade groups like FEMA (Flavor and Extract Manufacturers Association) to conduct safety evaluations through paid scientists, which raises concerns about impartiality.81 Consequently, it becomes easy for some companies, such as Prevagen, to exploit this loophole.82 Despite the FDA’s concerns about Prevagen’s active ingredient, Apoaequorin, and the product failing the FDA’s premarket review twice, the company successfully marketed the substance through Secret GRAS.83 This implies that food companies can also introduce any new flavors using this loophole to avoid addressing safety concerns, as seen in the usage of soy leghemoglobin in Impossible Beef.84 In granting it the GRAS status, the FDA conducted only a limited review, overlooking potential cancer risks associated with heme, which is present in red meat.85
The exploitation of the GRAS loophole also makes it hard for consumers to know what they are consuming. Specific ingredients in “natural flavors” are often not disclosed, leading consumers to assume “natural flavors” are solely composed of natural extracts.86 Consequently, this non-disclosure conceals health hazards due to consuming these flavors, which go through heavy chemical processing, have confirmed carcinogenic properties, and can come from unidentified plants and animals.87
B. BANNED AND UNSAFE FLAVORS
By law, the FDA has 180 days to respond to petitions of banning unsafe flavors, but oftentimes, a much longer time elapses with no response.88 One of the banned flavor ingredients, Methyleugenol, was classified as “reasonably anticipated to be a human carcinogen” by the National Toxicology Program (NTP) in 2002, and styrene had been under scrutiny since the 1970s, yet both were only banned around 2018.89 Despite styrene’s linkage to cancer, its ban only happened because public health groups fervently requested.90 Additionally, although the FDA first raised concerns about BVO’s safety in the 1970s, its use continued.91 It was only after decades of safety studies and pressure from public health advocates that the FDA finalized the ban in the past year to align with the U.K., E.U., and Japan.92
Compared to the E.U., the current ban of seven flavors in the U.S. is far from comprehensive. For example, the E.U. also bans capsaicin, which is responsible for spiciness in hot peppers, since pure capsaicin cannot be added to foods.93 Menthofuran and Quassin are also banned in the E.U. due to concerns about reproductive effects and liver damages that occur when consumed with alcoholic beverages.94 Furthermore, the U.S. has yet to systematically acknowledge that certain flavors, especially those in spices, are merely “undesirable” and do not arise to the level of prohibition.95 In other words, the E.U. varies its level of “ban” based on more factors other than a flavor’s carcinogenic property.
C. VAGUE LABELING
The FDA’s labeling rules have led to significant consumer confusion and ethical concerns, as flavor misbranding often involves vagueness in wording. First, there is the inadequate distinction between “natural” and “artificial” given the modern abundance of proprietary blends. The same chemical can appear as a natural flavor or artificial flavor depending on what it is made from. If vanillin comes from vanilla extract, it is labeled as “natural flavor” or “vanillin”; if it is synthesized in a lab, it can be labeled as “artificial flavor.”96 The FDA also allows natural flavors to be sourced from any natural substance, but does not mandate the naming of that substance.97 As a result, “natural beef flavor” can be from plants, where the “beef” describes the taste but not the source material.98 At the same time, conforming to specificity, such as printing “blueberry muffin with natural grape flavor”, is equally confusing.99 Consumers have trouble understanding these terms and are misled into thinking a product is purely natural or artificial, when flavors from genetically modified crops can be labeled “natural,” and natural flavors not derived from the expected characterizing flavor can be labeled “artificial.”100
There are also inconsistencies in flavor labeling court rulings, both in terms of how labels should be worded and how a reasonable consumer should understand them. In Lima, the court ruled that the use of honey imagery on Honey Bunches of Oats packaging was not misleading, since there was no explicit claim that honey was the primary sweetener.101 Conversely, in Viggiano, the court ruled that printing “all natural flavors” on diet soda, despite containing sucralose, would mislead consumers since they could not reasonably interpret “natural flavors” to apply to every ingredient in the product.102 The rulings for the two cases regarding water enhancer flavors were also at odds with one another. While for Target, the court dismissed the misleading claim, stating that a reasonable consumer would not expect a shelf-stable, brightly colored product to be free from artificial ingredients, it allowed the case to proceed for the MiO enhancers when it found that a reasonable consumer might believe “natural flavor” implies the absence of synthetic ingredients.103
Secondly, the current labeling system does not adequately address flavor allergens and “adjuvants.” While common allergen disclosure usually appear in a disclaimer, less common allergens or common-allergen-derived flavors (i.e. peanut-flavored milk) are be disclosed. For example, celery and coriander can be listed merely as “spice.”104 On the other hand, while flavor “adjuvants”, which add taste or aroma, and flavor “enhancers”, which modify flavors without introducing their own distinct taste, do not fall under “natural” or “artificial” flavors, the labeling of these substances are equally vague and even absent,105 leading flavor companies to erroneously categorize its taste modifiers as artificial flavors.
Third, animal-derived flavors raise an additional concern. Castoreum from beavers and ambergris from whales are often hidden behind the term “natural” flavor, which hurts consumers who seek to avoid animal products.106 While it is true that some animal-based ingredients, such as those derived from milk or crustacean shellfish, are subject to allergen labeling, many others, such as those from molluscan shellfish, can remain undetected as a result of the FDA’s lack of mandatory disclosure for these animal-derived substances.107
D. FDA ENFORCEMENT PROBLEMS
The FDA’s enforcement actions often fall short in effectiveness. Warning letters are often reactive and rely on voluntary company actions without guarantees for long-term compliance. Companies face no direct penalties upon receiving one, and unless widely publicized, these letters fail to alert consumers about potential risks associated with problematic products. Seizures and recalls only address products already in distribution, which means unsafe or mislabeled items may have already reached consumers before action is taken. While they may be effective on a case-by-case basis, they cannot address systemic non-compliances across industries and require significant FDA resources for investigations and legal approvals. Furthermore, ingredients and products are often sourced from multiple countries, and the FDA lacks the resources to inspect every shipment through import alerts and refusals.
Collaboration with the DOJ, while an important escalation tactic, often results in delayed actions. Civil penalties serve deterrent effects poorly since they are not consistently applied and may disadvantage smaller businesses when they are pinned against large companies with deep financial reserves that merely view fines as costs of doing business. Criminal prosecutions often require evidence of willful and egregious misconducts, and this allows many lesser but nonetheless severe violations, such as non-fraud cases, to fall through the cracks. Injunctions also require extensive legal proceedings and only come after repeated violations. The FDA’s inability to create new rules (as opposed to enforcing laws like NLEA and FALCPA) further limits its reach. Although rules like 21 C.F.R. § 101.22 mandate the identification of artificial and natural flavors, enforcement often depends on broad definitions that allow companies to obscure the presence of harmful ingredients under generic terms like “natural flavor.”
Finally, the lack of private action exacerbates these weaknesses, as individuals cannot initiate legal challenges directly under the FDA framework. This restriction leaves enforcement solely in the hands of the FDA, which has limited funding, resources, and inter-agency communications to monitor the growing food industry.
IV. POLICY RECOMMENDATIONS FOR THE FDA
A. CLOSING GRAS SELF-DETERMINATION LOOPHOLES
To address the loophole issue in the GRAS system, Congress should mandate that all new flavors undergo formal safety reviews instead of allowing self-determination of GRAS status. While a specific list of common substances could still qualify as GRAS, all new substances should be subject to premarket approval. Until then, through congressional legislations, companies should be mandated at the federal and state levels to submit compulsory notifications of all GRAS determinations, make full disclosures of flavors, and develop a central online database for food chemical usages. The FDA could also incorporate more independent third-party reviews into the GRAS notification process to ensure impartiality.
Mandating FDA pre-approval for all flavor chemicals could increase manufacturing costs, especially for small businesses. To mitigate these costs, Congress should consider a phased implementation for certain types of substances, mandating that the highest-risk flavors undergo FDA review while lower-risk or traditional ingredients are given extended compliance timelines. Congress could also offer incentives such as tax credits or grants to help cover the expenses for safety testing and third-party reviews. Specifically, such tax credits could equal to a percentage of the costs incurred during the FDA approval process. In the end, long-term benefits of increased consumer safety and confidence outweigh potential short-term costs for the industry.
B. SETTING MAXIMUM LEVELS FOR TOXIC SUBSTANCES
The FDA should follow the E.U.’s model and establish maximum allowable levels for a broader list of unsafe substances in both natural and artificial flavors. Specific maximum thresholds for toxic compounds that are commonly found in natural flavors should be set: for example, safrole (found in sassafras) can abide by the European food safety standards and coumarin (cinnamon, tonka beans) could be limited to no more than 2mg per kg of product to reduce potential liver toxicity at high levels.108 The FDA could protect consumers from overexposure by allowing for continued use in small, regulated quantities. This encourages manufacturers to reformulate products without eliminating traditional natural ingredients.
Furthermore, the FDA’s post-market monitoring for these safety quantities should be expanded to continuously reassess these substances. A mandatory periodic review cycle, every five years for example, for GRAS substances could be established. Alternatively, Congress could mandate a real-time adverse event reporting system for flavors specifically, where consumers, manufacturers and even healthcare providers would report adverse reactions directly to the FDA.109 This “centralized database” could in turn track unexpected allergies and in turn triggering immediate FDA review. Congress could also mandate a sampling and testing program.110 Like pesticide residue monitoring, this program would involve random sampling of food products to verify compliance, recalls of products exceeding limits, and penalties for repeated non-compliance. For industry stakeholders concerned about the burden on small farmers and spice growers, a similar phased compliance timeline can be introduced.
C. STRENGTHENING FLAVOR LABELING
Federal regulations should not allow manufacturers to obscure some ingredients behind vague catch-all terms such as “artificial” or “natural flavor.” The 21 C.F.R § 101.22 should be revised to mandate the full disclosure of flavor ingredients through product specification and label. Manufacturers should specify source materials to help consumers make informed choices, particularly those with dietary restrictions. For product specification, the FDA could mandate submissions of specification documents; already a critical component in quality assurance, such documents would list all sub-ingredients in flavor compounds and serve as an objective standard in cases of non-compliance. For instance, if a “natural flavor” contains traces of almond extract or other allergens, this information would be listed both in the spec and disclosed on the consumer label or accessible online via a QR code on packaging.111 The FDA could also introduce a tiered label review, which aims to catch not only hidden allergens, but also misbranding risks such as misleading “high fiber” or “antioxidant” claims.112 This label review process could also introduce additional disclosure statements for complex claims, such as stating “high in natural antioxidants” only when it meets a specified threshold.
To balance proprietary concerns and competitive advantages of products with transparency, the FDA could permit companies to group minor components or disclose only specific allergens and major ingredients. Alternatively, companies could be required to disclose all ingredients to the FDA confidentially with limited public access.
D. INCREASING RESOURCES FOR FDA ENFORCEMENT AND REVIEW
More funding should be allocated to the FDA to strengthen its ability to review food flavor petitions and monitor food safety post-market. The additional funding could reduce industry reliance on the GRAS loophole and encourage companies to seek formal approvals. However, increasing FDA funding may face budgetary challenges in Congress, as FDA resources should be focused on broader health concerns rather than niche flavor regulations. In this case, an alternative might be increasing user fees for companies submitting GRAS notifications or food flavor petitions, like the PDUFA (Prescription Drug User Fee Act) model for pharmaceuticals, but this is also at the risk of facing industry resistance and disadvantaging small businesses. Public-private partnerships between academic institutions and industry groups, and targeted federal research grants with other government agencies like the USDA, FEMA, and IOFI, may also help secure funding for specific projects, promote data and expertise sharing, and harmonize labeling standards across industries.113
V. CONCLUSION
In an increasingly complex food landscape, improvements in food flavors regulation are essential to ensuring consumer safety and maintaining compliance. It has become evident that current loopholes in the GRAS system, the use of toxic and poorly reviewed flavors, and vague labeling practices undermine the efficacy of existing regulations. To address these shortcomings, there must be a shift away from the current GRAS system, stricter controls on harmful flavors, higher uniformity in labeling standards, and more enhanced FDA oversight for these changes. The FDA aims to protect public health and restore trust in the food system; as the food industry continues to innovate, these proactive and adaptive measures are necessary to meet evolving complexities.
FOOTNOTES
- John Wright, Flavor Bites: Methyl Cyclopentenolone, PERFUMER & FLAVORIST (Nov. 30, 2018),
perfumerflavorist.com/flavor/ingredients/article/21856905/flavor-bites-methyl-cyclopentenolone. ↩︎ - Diacetyl, PERFUMERS WORLD, https://www.perfumersworld.com/view.php?pro_id=9DV00144 (last visited Oct. 20, 2024). ↩︎
- Kate Dwyer, The Truth About Natural and Artificial Flavors, BON APPETIT (Mar. 15, 2017),
bonappetit.com/story/truth-about-natural-artificial-flavors. ↩︎ - U.S. Department of Agriculture, Flavor Chemistry Research at the USDA Western Regional Research Center, AM. CHEMISTRY SOC’Y (Aug. 29, 2013), https://www.acs.org/education/whatischemistry/landmarks/usda-flavor-chemistry.html. ↩︎
- Id. ↩︎
- Id. ↩︎
- Id. ↩︎
- Julie Clark, Apple Pie with Nutmeg, TASTES OF LIZZY T, https://www.tastesoflizzyt.com/homemade-apple-pie/ (Aug. 10, 2024). ↩︎
- Tastykake Glazed Apple Pie Individually Wrapped Snack Pie, BAKERS PLUS, https://www.bakersplus.com/p/tastykake-glazed-apple-pie-individually-wrapped-snackpie/0002560000972 (last visited Oct. 22, 2024). ↩︎
- See id. ↩︎
- Reshna K.R., Sreerag Gopi & Preetha Balakrishnan, Introduction to flavor and Fragrance in Food Processing, AM. CHEMISTRY SOC’Y PUB. (Dec. 28, 2022), https://pubs.acs.org/doi/10.1021/bk-2022-1433.ch001. ↩︎
- Natural & Artificial Flavors: How Are They Different? FLAVORMAN (Apr. 21, 2021),
https://www.flavorman.com/media/natural-and-artificial-flavors-how-are-they-different [hereinafter Natural & Artificial Flavors]. ↩︎ - Beyond the Label: The Truth Behind “Natural Flavors”, LAIRD SUPERFOOD (Feb. 22, 2024),
https://lairdsuperfood.com/blogs/news/beyond-the-label-the-truth-behind-natural-flavors [hereinafter Beyond the Label]. ↩︎ - Natural & Artificial Flavors, supra note 12. ↩︎
- Beyond the Label, supra note 13 (Some of the most common natural compounds include amyl acetate (banana), linden ether (honey), and acetoin (butter)). ↩︎
- 21 C.F.R. § 501.22 (2024). ↩︎
- Natural & Artificial Flavors, supra note 12. ↩︎
- Id. ↩︎
- Id. ↩︎
- Id. ↩︎
- Natural Ingredients, DSM-FIRMIENICH, https://www.dsm-firmenich.com/en/businesses/perfumery-
beauty/ingredients/naturals.html (last visited Oct. 22, 2024). ↩︎ - Synthetic Ingredients, DSM-FIRMENICH, https://www.dsm-firmenich.com/en/businesses/perfumery-beauty/ingredients/synthetics.html (last visited Oct. 22, 2024). ↩︎
- See generally GEORGE A. BURDOCK, FENAROLI’S HANDBOOK OF FLAVOR INGREDIENTS (6th ed. 2009); see also John B. Hallagan, Richard L Hall & Joanna Drake, The GRAS provision—The FEMA GRAS Program and the Safety and Regulation of Flavors in the United States, 138 FOOD & CHEM. TOXICOLOGY 111236 (2020). ↩︎
- USDA v. FDA Food Labeling, EUROFINS, https://www.eurofinsus.com/food-testing/resources/usda-vs-fda-food-labeling/ (Jan. 5, 2024). ↩︎
- Id. ↩︎
- See generally FDCA, § 201(f); 21 U.S.C. § 321(f). ↩︎
- 21 U.S.C. § 342 (a). ↩︎
- 21 U.S.C. § 348; 21 C.F.R. § 170.3. ↩︎
- Staff Reporter, IFF Settles Claims with Lung-Damaged Popcorned Workers, BAKERY & SNACKS,
https://www.bakeryandsnacks.com/Article/2005/10/07/IFF-settles-claims-with-lung-damaged-popcorn-workers (July. 19, 2008, 4:24 PM); RICHARD FAMA, ESQ, BEWARE DIACETYL: THE NEXT LEGAL QUAGMIRE? 2 (2007); Kelly Frederick, The Future of Diacetyl, PERFUMER & FLAVORIST (Dec. 23, 2008), https://www.perfumerflavorist.com/flavor/regulatory-research/news/21870700/the-future-of-diacetyl. ↩︎ - See Nutrition Labeling and Education Act of 1990, Pub. L. No. 101-535, 104 Stat. 2353 (1990). ↩︎
- Id. ↩︎
- See Food Allergen Labeling and Consumer Protection Act of 2004, Pub. L. No. 108-282, §201, 118 Stat. 905, 905-911 (2004). ↩︎
- 21 U.S.C. § 321 (s); Food Additives Amendment of 1958, Pub. L. No. 85-929, 72 Stat. 1784 (1958); Paulette M. Gaynor et al., FDA’s Approach to the GRAS Provision: A History of Processes, FDA (Apr. 2006), https://www.fda.gov/food/generally-recognized-safe-gras/fdas-approach-gras-provision-history-processes# ↩︎
- 21 U.S.C. § 348. ↩︎
- 21 U.S.C. § 348. ↩︎
- U.S. FOOD & DRUGS ADMIN., GUIDANCE FOR INDUSTRY: QUESTIONS AND ANSWERS ABOUT THE FOOD ADDITIVE OR COLOR ADDITIVE PETITION PROCESS (2011). ↩︎
- Id. ↩︎
- Food Additives Amendment of 1958, Pub. L. No. 85-929, 72 Stat. 1784 (1958). ↩︎
- FDCA, § 201(f); 21 U.S.C. § 321(s); 21 C.F.R. § 170, 182; Gaynor et al., supra note 30. ↩︎
- Id. (The scientific confirmation demonstrates the nature of the substances, conditions of use, and information available to scientists). ↩︎
- Id. ↩︎
- Secret Ingredients Lurk in Food, Hidden as ‘Artificial Flavor,’ ‘Natural Flavor,’ and ‘spices’, CTR. FOR SCI. PUB. INT., https://www.cspinet.org/press-release/secret-ingredients-lurk-food-hidden-artificial-flavor-natural-flavor-and- spices (Mar. 19, 2024) [hereinafter Secret Ingredients Lurk]. ↩︎
- Id. ↩︎
- Id. ↩︎
- Id. ↩︎
- Id. ↩︎
- 21 C.F.R. § 172.515, 189.180. ↩︎
- See U.S. FOOD & DRUGS ADMIN., FDA REMOVES 7 SYNTHETIC FLAVORING SUBSTANCES FROM FOOD ADDITIVES LIST (2018). ↩︎
- Id. ↩︎
- Healthday, FDA Bans Food Additive Found in Sports Drinks, Sodas, U.S. NEWS (July 3, 2024),
https://www.usnews.com/news/health-news/articles/2024-07-03/fda-bans-food-additive-found-in-sports-drinks-sodas. ↩︎ - Id. ↩︎
- See discussion infra Sections III.B. ↩︎
- See Mɪɴɴ. Dᴇᴘ’ᴛ. Hᴇᴀʟᴛʜ, Tᴏxɪᴄᴏʟᴏɢɪᴄᴀʟ Sᴜᴍᴍᴀʀʏ ғᴏʀ: Bᴇɴᴢᴏᴘʜᴇɴᴏɴᴇ (2023); Ethyl Acrylate, Pᴜʙ. Cʜᴇᴍ, https://pubchem.ncbi.nlm.nih.gov/compound/Ethyl-acrylate; Xiaoling Zhang et al., A Cautionary Tale for Using Read-across for Cancer Hazard Classification: Case Study of Isoeugenol and Methyl Eugenol, 136 Rᴇɢᴜʟ. Tᴏxɪᴄᴏʟᴏɢʏ & Pʜᴀʀᴍᴀᴄᴏʟᴏɢʏ 105280 (2022); Nᴀᴛ’ʟ. Tᴏxɪᴄᴏʟᴏɢʏ Pʀᴏɢ., Rᴇᴘᴏʀᴛ ᴏɴ Cᴀʀᴄɪɴᴏɢᴇɴs: Sᴛʏʀᴇɴᴇ (15th ed.); Iɴᴛ’ʟ. Aɢᴇɴᴄʏ. Rsᴄʜ. Cᴀɴᴄᴇʀ, Sᴏᴍᴇ Cʜᴇᴍɪᴄᴀʟs Tʜᴀᴛ Cᴀᴜsᴇ Tᴜᴍᴏʀs ᴏғ ᴛʜᴇ Uʀɪɴᴀʀʏ Tʀᴀᴄᴛ ɪɴ Rᴏᴅᴇɴᴛs Vᴏʟᴜᴍᴇ 119 (2017); Myrcene, SᴄɪᴇɴᴄᴇDɪʀᴇᴄᴛ (2020), https://www.sciencedirect.com/topics/pharmacology-toxicology-and-pharmaceutical-science/myrcene; Verena Voigt, Heike Franke & Dirk W. Lachenmeier, Risk Assessment of Pulegone in Foods Based on Benchmark Dose–Response Modeling, 13 Fᴏᴏᴅs 2906 (2024), https://doi.org/ 10.3390/foods13182906. ↩︎
- Jennifer Allen, Food Labeling Requirements for Natural Flavors, FOOD SAFETY TECH (Nov. 28, 2023), https://foodsafetytech.com/column/food-labeling-requirements-for-natural-flavors/. ↩︎
- Class Action Alert—Characterizing Flavor, FOOD L. FIRM (Feb. 5, 2024), https://www.foodlawfirm.com/chewed-up/class-action-alert-characterizing-flavor-label-review-natural-artificial-fda/ [hereinafter Class Action Alert]. ↩︎
- Allen, supra note 54. ↩︎
- Class Action Alert, supra note 55. ↩︎
- Id. ↩︎
- Id. ↩︎
- Id. ↩︎
- Id. ↩︎
- Id; U.S. FOOD & DRUGS ADMIN., WHAT’S IN A NAME? WHAT EVERY CONSUMER SHOULD KNOW ABOUT FOODS AND FLAVORS (2016) (for example, if a maple food is made with maple syrup or a maple flavor comes from a natural maple flavor, one may see” maple syrup” or “natural maple syrup” directly in the ingredients list.) ↩︎
- FDCA, 21 U.S.C. § 352 (2024); 21 C.F.R. § 101.18. ↩︎
- Christopher P. Fitzgerald, Increased Litigation Regarding Food and Beverage Product Labeling Creates Risk for Manufacturers, CONN KAVANAUGH, https://www.connkavanaugh.com/new-england-product-liability-insights-and-resources/increased-litigation-regarding-food-and-beverage-product-labeling-creates-risk-for-manufacturers/ (last visited Oct. 22, 2024). ↩︎
- 944 F.Supp.2d 877, 881 (C.D. Cal. 2013). ↩︎
- Kaixin Fan & Ashley Simonsen, Two Federal Courts Arrive at Opposite Conclusions in Suits Claiming “Natural Flavor with Other Natural Flavors” is Misleading, COVINGTON (Jan. 18, 2023),
https://www.insideclassactions.com/2023/01/18/two-federal-courts-arrive-at-opposite-conclusions-in-suits-claiming-natural-flavor-with-other-natural-flavors-is-misleading/ ↩︎ - Dwyer, supra note 3. ↩︎
- 21 C.F.R. 172.510. ↩︎
- Id; see 21 C.F.R. 582.60. ↩︎
- Id. ↩︎
- See discussion infra Section III.C. ↩︎
- U.S. FOOD & DRUGS ADMIN., WARNING LETTER TO MIDWEST FOOD, LLC, REF NO. 677464 (2024). ↩︎
- 73 U.S. FOOD & DRUGS ADMIN., WARNING LETTER TO LIPARI FOODS OPERATING COMPANY, LLC, REF NO. 669083 (2024). ↩︎
- U.S. FOOD & DRUGS ADMIN., HOW SAFE ARE COLOR ADDITIVES (2023). ↩︎
- See Healthday, supra note 50. ↩︎
- See U.S. FOOD & DRUGS ADMIN., IMPORT ALERT FOR INDUSTRY SPICES, FLAVORS AND SALTS. ↩︎
- See U.S. FOOD & DRUGS ADMIN., ECONOMICALLY MOTIVATED ADULTERATION (FOOD FRAUD) (2024) [hereinafter ECONOMICALLY MOTIVATED ADULTERATION]. ↩︎
- Id. (Casey’s Seafood did a bait-and-switch with foreign crab meat and Castle Cheese added wood pulp and cheddar to grated parmesan while selling as 100% parmesan cheese). ↩︎
- U.S. FOOD & DRUGS ADMIN., HOW U.S. FDA’S GRAS NOTIFICATION PROGRAM WORKS (2006). ↩︎
- Secret Ingredients Lurk, supra note 42. ↩︎
- See Id. ↩︎
- See Chiara Eisner, Americans Took Prevagen for Years—as the FDA Questioned Its Safety, WIRED (Oct. 21, 2020, 7:00 AM), https://www.wired.com/story/prevagen-made-millions-fda-questioned-safety/. ↩︎
- Id. ↩︎
- See generally IMPOSSIBLE FOODS INC., GRAS NOTIFICATION FOR SOY LEGHEMOGLOBIN PROTEIN PREPARATION DERIVED FROM PICHIA PASTORIS (2018). ↩︎
- Raychel E. Santo et al., Considering Plant-Based Meat Substitutes and Cell-Based Meats: A Public Health and Food Systems Perspective, FRONTIERS (Aug. 30, 2020), https://www.frontiersin.org/journals/sustainable-food-systems/articles/10.3389/fsufs.2020.00134/full ↩︎
- Beyond the Label, supra note 13. ↩︎
- Id. ↩︎
- 21 U.S.C. § 348; Ben Roussel, Advocates Sue FDA to Force Decision on Cancer-Causing Chemicals in Popular Foods, EARTH JUST. (May 2, 2018), https://earthjustice.org/press/2018/advocates-sue-fda-to-force-decision-on-cancer-causing-chemicals-in-popular-foods. ↩︎
- See generally U.S. DEP’T HEALTH & HUM. SERVS., METHYLEUGENOL, STYRENE, in REPORT ON CARCINOGENS (15th ed. 2021). ↩︎
- Id. ↩︎
- See Healthday, supra note 50. ↩︎
- Id. ↩︎
- EUR. FOOD SAFETY AUTH., OUTCOME OF THE CONSULTATION WITH MEMBER STATES AND EFSA ON THE BASIC SUBSTANCE APPLICATION FOR APPROVAL OF CAPSICUM ANNUUM L. VAR. ANNUUM, LONGUM GROUP, CAYENNE EXTRACT TO BE USED IN PLANT PROTECTION AS A REPELLENT TO SEED-EATING MAMMALS AND BIRDS (2020). ↩︎
- See generally M. de Nijs, M. Y. Noordam & H. G. J. Mol, Short Inventory of EU Legislation on Plant Toxins in Food, 9 QUALITY ASSURANCE & SAFETY OF CROPS & FOOD 129 (2016). ↩︎
- See generally id. ↩︎
- Thomas Galligan & Adrienne Crezo, What’s in “Flavor”? No One Knows—and That’s a Problem, CTR. FOR SCI. PUB. INT., https://www.cspinet.org/cspi-news/whats-flavor-no-one-knows-and-thats-problem (Mar. 12, 2024). ↩︎
- Id. ↩︎
- Id. ↩︎
- See Allen, supra note 54. ↩︎
- Beyond the Label, supra note 13. ↩︎
- Fitzgerald, supra note 64. ↩︎
- 944 F.Supp.2d 877, 881 (C.D. Cal. 2013). ↩︎
- Fan & Simonsen, supra note 66. ↩︎
- Susan L. Hefle, Julie A. Nordlee & Steve L. Taylor, Allergenic Foods, 36 CRITICAL REVS. FOOD SCI. & NUTRITION S69, S74 (1996). ↩︎
- 21 C.F.R. 172.510. ↩︎
- 21 C.F.R. 182.50 (certain other spices, seasonings, and natural extracts). ↩︎
- Id. ↩︎
- See generally Mirjana Loncar et al., Coumarins in Food and Methods of Their Determination, 9
MULTIDISCIPLINARY DIGIT. PUBL’G. INST. 645 (2020). ↩︎ - See, e.g., U.S. FOOD & DRUGS ADMIN., CFSAN ADVERSE EVENT REPORTING SYSTEM (CAERS) (2024) (The FDA already has a similar system in place for foods, dietary supplements and cosmetics. Having a separate one for food flavors specifically may be useful for safety monitoring). ↩︎
- ECONOMICALLY MOTIVATED ADULTERATION, supra note 77 (The FDA has done such sampling assignments in the past for detecting Sudan dyes, which can cause cancer and are illegal for food use, in imported palm oil). ↩︎
- Product Specifications: The Critical Document for a Food Business, Fᴏᴏᴅ L. Fɪʀᴍ (Dec. 20, 2023),
https://www.foodlawfirm.com/chewed-up/product-specification-the-most-important-document-to-a-food-business/. ↩︎ - FDA Food Labeling Review and Compliance, Fᴏᴏᴅ L. Fɪʀᴍ, https://www.foodlawfirm.com/what-we-do/food-labeling-compliance/ (last visited Oct. 22, 2024). ↩︎
- For example, while the USDA allows “flavor” generally, the FDA currently requires specific terms like “natural flavor”. USDA also mandates disclosing an added smoked flavor, but FDA does not. Aligning these standards would reduce consumer confusion and generate consistency. ↩︎